The molecular formula HCOOCH CH2 H2O might not be as well-known as H₂O or CO₂. The sort of thing I love about this compound is how it plays a central role in organic chemistry’s reaction pathways. This combination brings together methyl formate (HCOOCH), a methylene group (CH2), and water (H₂O). These components work together in the most important chemical processes.
Chemists typically focus on methyl formate’s hydrolysis, shown by the balanced equation HCOOCH₃ + H₂O → HCOOH + CH₃OH. This reaction creates formic acid (HCOOH), which stands as the simplest carboxylic acid. The global production reached 720 thousand tons per year in 2009. Formic acid shows high reactivity because of its carboxylic acid group. It proves about ten times stronger than acetic acid and has a dissociation constant (pKa) of 3.745.
The H₂O displayed formula helps chemists predict molecular behavior before lab work begins. These chemical relationships go beyond the lab setting. Formic acid proves valuable in leather tanning, rubber coagulation, and preservation. Scientists now see it as a promising hydrogen source to propel development in fuel cells.
Decoding the Formula: What HCOOCH CH2 H2O Represents
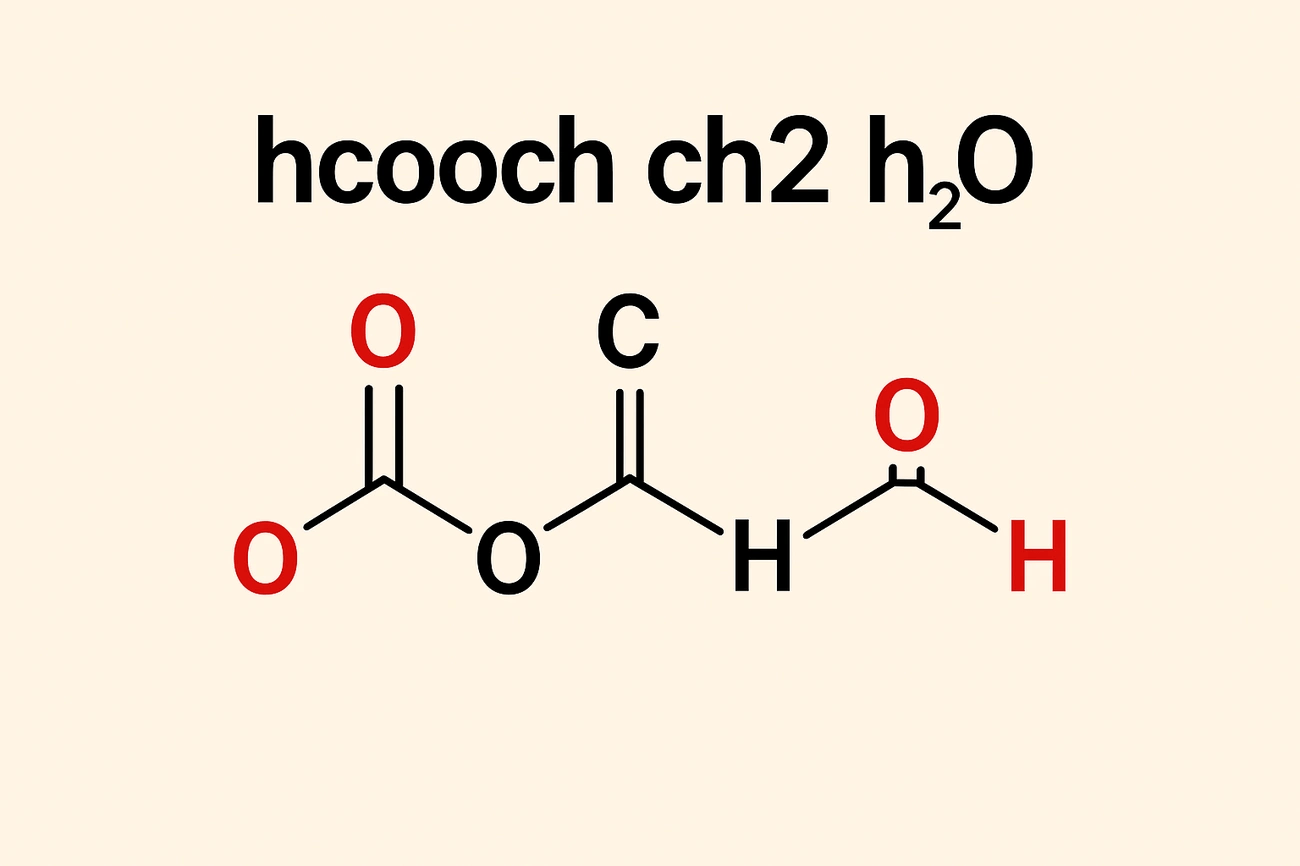
Image Source: Howey Industries
HCOOCH CH2 H2O looks complex at the molecular level but plays a vital role in organic chemistry. A closer look at this notation shows how individual components interact with each other instead of forming one molecule.
Understanding the Ester, Methylene, and Water Components
HCOOCH represents methyl formate – a simple ester that forms from formic acid (HCOOH) and methanol (CH₃OH). The structural formula HCOOCH₃ shows a formyl group (HCOO-) bonded to a methyl group (-CH₃). This colorless liquid has a sweet, ether-like smell and works as a solvent and intermediate in chemical manufacturing. Methyl formate evaporates quickly with its low boiling point of 31.5°C, making it perfect to use in fast-drying processes.
CH₂ identifies the methylene group – a reactive carbon fragment with two hydrogen atoms. While it can’t exist alone, methylene is a key building block in organic synthesis and polymer chemistry. This reactive unit connects organic molecules and streamlines processes in hydrocarbon chains and polymers. CH₂ units show up in biological systems like lipids, proteins, and nucleic acids to help build membrane structure and store energy.
Water or H₂O helps many chemical reactions happen as the universal solvent. It dissolves many substances because of its polar nature. Water’s hydrogen bonds give it high melting and boiling points unusual for its size. Water acts as both reactant and medium in reactions with esters like methyl formate, especially during hydrolysis.
Why It’s Not a Single Molecule but a Reaction System
People often think HCOOCH CH2 H2O is one compound. The notation actually describes a reaction system of components in methyl formate’s hydrolysis, where water breaks down the ester bond.
The main reaction looks like this:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
Methyl formate reacts with water to create formic acid (HCOOH) and methanol (CH₃OH). The reaction needs specific conditions to work well, often using acid or base catalysts to make the ester bond react better with water. Heat can make the reaction happen faster.
Water molecules gather around the polar HCOOCH group in solution. The methylene parts can join reaction pathways and create complexes that accelerate chain growth or breaking. These interactions show how the components work together rather than existing as one structure.
This reaction system matters beyond chemistry labs. Formic acid from this process helps in leather processing and agriculture. Methanol serves as a basic solvent and fuel component. On top of that, it shows how esterification can reverse – something we use in chemical recycling and organic synthesis.
HCOOCH CH2 H2O shows how formic acid (HCOOH), methylene bridge (CH₂), and water (H₂O) work together. These components are the foundations of many organic syntheses, redox processes, and new fuel-cell technologies. The sort of thing I love is how these chemical parts interact to power important industrial and biological processes, even though they don’t make a single stable molecule.
How to Read and Interpret Organic Formulas
Image Source: Chemistry Steps
Reading organic formulas needs a good grasp of molecular notation—a system of symbols and numbers that shows vital structural information. A chemist must figure out each character and subscript to understand why chemical relationships happen in compounds like HCOOCH CH2 H2O.
Subscripts and Bonding Patterns in CH₂ and H₂O
Chemical symbols with subscripts make up a molecular formula to show atom types and quantities in a molecule. The subscript immediately follows the element symbol and tells us how many atoms exist. A water molecule (H₂O) has two hydrogen atoms shown by the subscript “2”, while oxygen has no subscript because there’s just one atom.
The difference between a subscript after a symbol and a number before it matters. H₂ shows a diatomic hydrogen molecule where two hydrogen atoms bond together. 2H means two separate hydrogen atoms. This means 2H₂ would show two molecules of diatomic hydrogen.
CH₂ plays a vital role in organic chemistry. Unlike H₂O (a stable molecule), CH₂ rarely exists alone. This group usually connects larger organic structures as a methylene group. Carbon makes four bonds—two with hydrogen atoms and two with other atoms or groups. CH₂ sometimes refers to methylene carbene, a highly reactive species where carbon has two unshared electrons.
The methylene group (-CH₂-) builds most hydrocarbons by creating chains in compounds like hexane (C₆H₁₄) or octane (C₈H₁₈). These -CH₂- unit chains are the foundations of many organic compounds, including those in the HCOOCH CH2 H2O system.
H₂O’s bonding pattern shows a molecule where oxygen’s higher electronegativity creates polarity. Water molecules can dissolve ionic and polar substances well because of this trait. Atoms in water connect through covalent bonds, sharing electrons to complete each atom’s outer shell.
Rules to read subscripts in organic formulas:
- Subscripts only affect the element right before them
- Use subscripts only for multiple atoms
- No subscript means one atom
- Parentheses group atoms before applying subscripts
H₂O Displayed Formula in Reaction Contexts
Water’s formula (H₂O) means different things in reactions. It can work as a solvent, product, or reactant.
Water aids reactions as a solvent by letting ions and molecules mix better. Condensation reactions, like making esters, produce water. Water breaks down bigger molecules during hydrolysis reactions as a reactant.
Water on the product side often points to a condensation reaction in HCOOCH CH2 H2O reactions. Two molecules join and lose a water molecule. This happens often in making esters, ethers, or polymers.
The balanced equation for methyl formate (HCOOCH₃) hydrolysis shows:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This equation shows water acting as a nucleophile in acid-catalyzed hydrolysis. It attacks the ester group’s electrophilic carbon. A tetrahedral intermediate forms and yields formic acid (HCOOH) and methanol (CH₃OH).
Chemists think over stoichiometry—how much reactant and product we need. H₂O on either side means counting hydrogen and oxygen atoms to balance the reaction. This helps predict how atoms move during molecular changes.
These notation rules let chemists read organic formulas like a language. They can predict how molecules behave and reactions end before testing. Becoming skilled at reading subscripts, bonding patterns, and displayed formulas helps chemists succeed in organic synthesis and analysis.
Reactions Involving HCOOCH CH2 H2O in Organic Chemistry
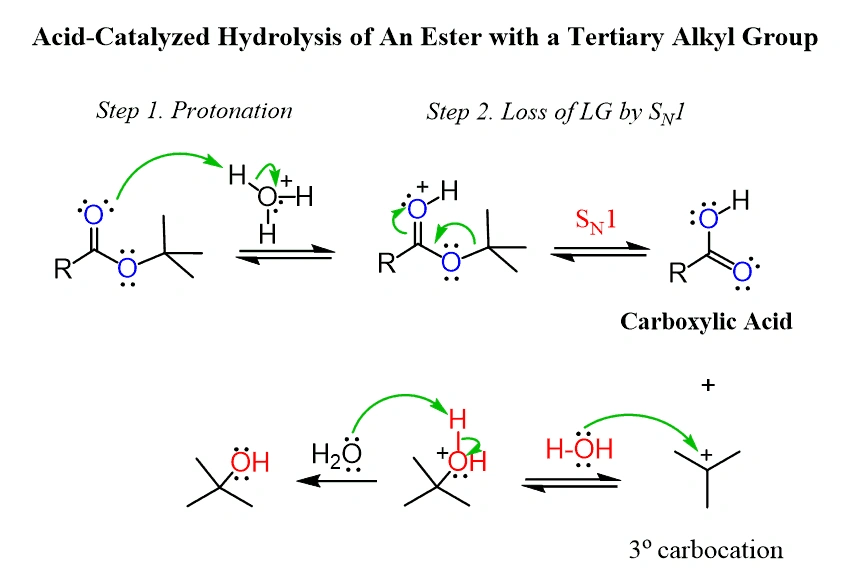
Image Source: Chemistry Steps
The chemical interaction between methyl formate, methylene groups, and water creates a captivating array of reaction pathways that are the life-blood of many organic synthesis procedures. These reactions show the basic principles of how molecules change and interact under different conditions.
Hydration and Hydrolysis Pathways
The HCOOCH CH2 H2O system’s most important reaction is hydrolysis. Water breaks down methyl formate (HCOOCH₃) to produce formic acid and methanol according to the balanced equation:
HCOOCH₃ + H₂O → HCOOH + CH₃OH
This change shows how esters turn back into their carboxylic acid and alcohol components when water cleaves their bonds. The reaction takes different paths based on the conditions used.
The mechanism starts with protonation of the carbonyl oxygen in methyl formate under acidic conditions. This increases the electrophilic character of the carbon atom. A water molecule then attacks this carbon as a nucleophile and forms a tetrahedral intermediate. The ester bond breaks after proton transfer. This releases methanol and creates protonated formic acid, which then deprotonates to create the neutral acid molecule.
Basic conditions (saponification) allow hydroxide ions to attack the ester directly without protonation. This path first creates formate ions and methanol. These must become neutral before formic acid can form. Industrial applications prefer acidic conditions because they make product separation cleaner and improve yields.
Organic chemistry education uses methyl formate hydrolysis to teach nucleophilic substitution, acid-base catalysis, and molecular stability principles. This reaction helps students understand how stable compounds can change under controlled conditions.
Esterification Reversibility and Water’s Role
Esterification reverses hydrolysis. Carboxylic acids react with alcohols to create esters and water. This reversible process follows the general equation:
RCOOH + R’OH ⇌ RCOOR’ + H₂O
For methyl formate specifically:
HCOOH + CH₃OH ⇌ HCOOCH₃ + H₂O
The double arrow (⇌) shows that this reaction reaches equilibrium unless conditions push it one way or the other. This reversibility makes esterification perfect for studying chemical equilibria.
Water serves several key functions in these reactions:
- Active Reactant: Water acts as a nucleophile that attacks the ester group’s electrophilic carbon and breaks the ester bond during hydrolysis.
- Product: Esterification produces water, and removing it can drive the reaction forward according to Le Chatelier’s principle.
- Reaction Medium: Polar reactants interact better in water, which also stabilizes transition states through hydrogen bonding.
- pH Regulator: Water helps maintain proper pH levels in aqueous solutions, which affects reaction rates and yields substantially.
Chemists use several methods to push esterification forward against its natural reversibility:
- They use excess alcohol or acid to shift equilibrium
- They remove water as it forms through distillation or molecular sieves
- They add concentrated sulfuric acid as a catalyst and dehydrating agent
- They apply heat with reflux conditions for larger esters that form more slowly
Adding extra water pushes the equilibrium toward hydrolysis. This lets chemists control reaction direction by adjusting water concentration. Biodiesel production, pharmaceutical synthesis, and flavor compound manufacturing use this principle.
The HCOOCH CH2 H2O reaction system demonstrates eco-friendly chemistry principles. Water provides an environmentally safe medium for chemical transformations. These water-based processes create fewer toxic byproducts than reactions in organic solvents, which supports sustainable chemistry goals.
Real-World Applications of the Reaction System
Image Source: ACS Publications – American Chemical Society
The reaction components of the HCOOCH CH2 H2O system do more than just exist in theoretical chemistry – they’re the foundation for many industrial processes and consumer products. These practical applications show how basic chemical interactions create real value in many different sectors.
Formic Acid in Agriculture and Energy
Formic acid, which comes from methyl formate hydrolysis, plays a vital role in modern agriculture. Farmers commonly use it to preserve livestock feed, where its acidity inhibits bacterial growth, including dangerous bacteria like Escherichia coli. This preservation makes feed last longer and helps large-scale animal operations save money. The acid also helps make better silage by promoting lactic acid fermentation while stopping butyric acid from forming, which results in nutrient-rich animal feed.
Farmers also use formic acid to manage pests. It works as both a pesticide and herbicide to protect crops from unwanted pests and weeds. Beekeepers use it to curb parasitic Varroa mites in their colonies without making the honey unsafe.
Formic acid has become one of the most promising and safest ways to generate hydrogen under mild conditions. This makes it an excellent energy carrier. When it breaks down in a controlled way, formic acid releases hydrogen gas, providing a safe, dense hydrogen source for fuel cells. Small electronic devices and portable power systems can run on direct formic acid fuel cells (DFAFCs) that use liquid formic acid as fuel.
A single kilogram of formic acid can store about 33 kWh of energy, which simple, reliable technologies can extract. This capability has led to new ways of using formic acid made from carbon dioxide as a green energy storage solution.
Methanol in Biodiesel and Solvent Systems
Methanol, another product from our reaction system, is essential for making biodiesel. It’s the most common alcohol used in transesterification reactions, making up roughly 20% of biodiesel’s weight. The production usually involves methanol reacting with oils using a catalyst.
The environmental impact of biodiesel production changes based on where the methanol comes from. Research shows that making methanol from coke oven gas and carbon dioxide, then using it for biodiesel production with alkali catalysis, is best for the environment. Using biomass as a renewable source makes the environmental benefits even better in certain areas.
Methanol-based transesterification reactions can be very effective. Scientists have achieved 94% yields under the right conditions (methanol-to-oil ratio of 6:1, 0.5 wt% catalyst, 60°C, and 2-hour reaction time). High-frequency ultrasound methods can speed this up, reaching over 90% yield in just 30 minutes.
Beyond biodiesel, methanol serves as a key industrial chemical. It helps make formaldehyde, acetic acid, and works as a fuel additive. Its relatively safe nature and ability to carry hydrogen make it valuable for environmentally friendly applications.
CH₂ in Plastics and Synthetic Fibers
The methylene group (CH₂) might not exist on its own, but it’s the building block for many synthetic materials. Polymers with CH₂ units are the foundations for plastics, coatings, and fibers.
Polyethylene, a polymer made of repeated CH₂ units, ranks among the most used materials for packaging, containers, bags, and films. These products keep goods safe from moisture and contamination while staying light and strong. Polypropylene, another polymer rich in CH₂, goes into car parts like dashboards and bumpers.
The textile industry relies heavily on methylene-based chemistry. Synthetic fibers such as polyester (polyethylene terephthalate or PET), nylon, rayon, acrylic, and spandex all contain CH₂ groups in their structure. These materials are strong, stretchy, and resist environmental damage, making them perfect for clothes and industrial textiles.
CH₂-containing polymers also work well in electronics as insulating materials and device housings. Medical applications use biodegradable polymers like polylactic acid (PLA) and polycaprolactone (PCL), which contain methylene groups, to make sutures, implants, and controlled-release drug formulations.
These synthetic CH₂-containing materials are useful but pose environmental challenges. Unlike natural organic materials, synthetic polymers in plastics can last for centuries with almost no changes. This durability shows why we need to use and dispose of these valuable materials responsibly.
Using HCOOCH CH2 H2O as a Teaching Tool
The HCOOCH CH2 H2O system serves as an excellent teaching tool in chemistry classrooms worldwide. Students often work with this combination as they learn simple organic chemistry concepts. This exposure helps them grasp more complex molecular interactions.
Pattern Recognition in Organic Chemistry
Chemistry teachers use the HCOOCH CH2 H2O system to teach ester hydrolysis, nucleophilic substitution reactions, and functional group identification. This chemical combination makes a perfect starting point to teach pattern recognition because it has multiple functional groups that interact in predictable ways. Students learn to spot similar patterns across different molecular systems by analyzing how methyl formate interacts with water.
The hydrolysis of HCOOCH₃ may seem simple, but it reveals deeper concepts about reaction mechanisms, electrophilic attack, nucleophilic substitution, and transition states. Students who understand this reaction system can easily spot similar patterns in more complex organic molecules. These pattern recognition skills are a great way to get ready for advanced topics like polymer chemistry, biochemistry, and green chemistry.
Students often find this chemical system hard to understand without proper structural diagrams. Teachers help overcome this challenge by asking students to draw structural formulas and write hydrolysis equations. These exercises build vital molecular visualization skills that apply to many other chemical systems.
Predicting Products from Functional Groups
The HCOOCH CH2 H2O system shows how functional groups shape reaction pathways and products. Students who grasp the simple esterification and hydrolysis mechanisms can predict outcomes in similar reactions. This skill forms the foundation of organic chemistry.
Lab experiments show how water can act as a reactant, product, or solvent. This knowledge helps students predict reaction outcomes in different contexts. Understanding this basic reaction system improves their grasp of organic chemistry fundamentals and enhances their skills in industrial synthesis and laboratory research.
Chemistry teachers find this system helpful to explain organic reactions, bonding patterns, and hydration effects. Students learn about reactions like HCOOCH₃ + H₂O → HCOOH + CH₃OH and develop the analytical framework needed for more challenging reaction mechanisms in their chemistry education.
Common Misconceptions and Clarifications
People often misunderstand HCOOCH CH2 H2O in chemistry discussions. These misconceptions create confusion about how this reaction system works and behaves.
Is CH₂ a Stable Molecule?
CH₂ (methylene) is not a stable standalone molecule. Scientists can only detect this highly reactive carbene at very low temperatures or as a brief intermediate in chemical reactions. Methylene turns into excited oligomers through spontaneous autopolymerization, which makes isolation almost impossible under normal conditions. CH₂ typically works as a reactive group within larger molecular structures. Methylene’s unpaired valence electrons sit in separate atomic orbitals with independent spins in its lowest energy state—scientists call this the triplet state.
Does HCOOCH CH2 H2O Form a Single Compound?
Many people believe that “hcooch ch2 h2o” makes up a stable compound or direct reaction product. This notation does not refer to a single molecule. Scientists cannot find this molecule in databases like PubChem or Reaxys. The notation shows a reactive system that has methyl formate, a methylene-containing species, and water. Spaces between these components indicate separate parts that interact through reactions rather than forming one structure.
How to Get H₂O in Reactions: Reactant vs Solvent
Water plays many vital roles in these chemical processes. It works as both a solvent and reagent. Water helps stabilize ions during bond rearrangements as a solvent. It also breaks down complex molecules into simpler ones through hydrolysis as a reactant. The reaction rates depend on water’s temperature and pH control. Many organic reactions would not achieve proper energetic or kinetic balance without water.
Conclusion
The HCOOCH CH2 H2O system gives us a great way to understand the basic mechanisms of organic chemistry. These components interact with each other rather than existing as one structure. The notation shows methyl formate, a methylene group, and water – elements that take part in major chemical reactions like hydrolysis and esterification.
When methyl formate undergoes hydrolysis with water, it produces formic acid and methanol. This process shows us the basic principles of organic reactivity. Understanding the subscripts and bonding patterns in this formula helps predict how molecules behave in different chemical scenarios. Water is quite versatile – it can act as a reactant, product, or solvent based on the reaction’s context.
The real-life application of this chemical system is remarkable. Industries use formic acid from these reactions in agriculture, food preservation, and promising fuel cell technologies. Methanol plays a vital role in biodiesel production and works as an industrial solvent. The methylene groups are the foundations of many polymers used to make plastics and synthetic fibers.
Chemistry teachers worldwide use this system to help students recognize reaction patterns and predict outcomes based on functional group behavior. Students can apply these skills to more complex chemical systems they’ll encounter in their education and careers.
Let’s clear up some common misunderstandings about this chemical notation. CH₂ isn’t a stable molecule – it’s a reactive group within larger structures. HCOOCH CH2 H2O represents interactive components instead of a single compound. Water does more than just act as a solvent.
The HCOOCH CH2 H2O system shows how chemical notations help us tap into the full potential of molecular interactions. These chemical principles shape the technologies we need in modern life, from lab reactions to industrial uses. Understanding such systems helps chemists tackle challenges in fields from pharmaceuticals to green energy, showing how basic chemical knowledge propels innovation forward.
FAQs
1. What does HCOOCH CH2 H2O represent in organic chemistry?
HCOOCH CH2 H2O represents a reaction system involving methyl formate (HCOOCH), a methylene group (CH2), and water (H2O). It’s not a single molecule, but rather components that interact in important chemical processes.
2. How does the hydrolysis of methyl formate occur?
The hydrolysis of methyl formate occurs when water breaks down the ester bond. The reaction is typically represented as HCOOCH₃ + H₂O → HCOOH + CH₃OH, producing formic acid and methanol.
3. What are some real-world applications of formic acid?
Formic acid, produced from this reaction system, has various applications including use as a preservative in livestock feed, as a pesticide in agriculture, and as a promising hydrogen source for fuel cells in the energy sector.
4. Why is the HCOOCH CH2 H2O system useful as a teaching tool?
This system is valuable for teaching organic chemistry concepts like ester hydrolysis, nucleophilic substitution, and functional group identification. It helps students develop pattern recognition skills and the ability to predict products from functional groups.
5. Is CH₂ a stable molecule on its own?
No, CH₂ (methylene) is not a stable standalone molecule. It exists primarily as a highly reactive carbene or as a short-lived intermediate in chemical reactions. In organic compounds, it typically functions as a reactive group within larger molecular structures.

